PHYSICAL SCIENCES
CHEM-1010 Intro to Chemistry
My signature assignment is an essay on electrochemistry.
Reflective Writing: I have always wanted to have an understanding of basic chemical properties. Chemistry is the building block of all matter through out the universe and now I have an understanding of how it was formed and the relation between the elements and compounds. This is not an easy class, however I think the objectives focus on the fundamentals that give an overall basic knowledge of chemistry that everyone should know. Through out the text I recognized many of the chemicals that surround modern life. Chem-1010 is an essential part of education and life.
CHEM-1010 Intro to Chemistry
Electrochemistry
By: Michael Barela
Electrochemistry is the study of reactions in which charged particles (ions or electrons) cross the interface between two phases of matter, typically a metallic phase (the electrode) and a conductive solution, or electrolyte. This chemical reaction generally known as an electrode process is also called electrode reactions.
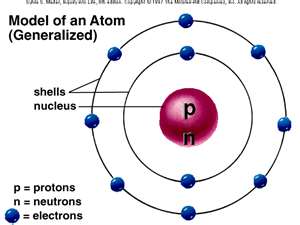
In the fourth century B.C. Greek philosophers believed that matter was composed of indestructible building blocks that were so small that they could not be seen. They called these building blocks atomos, meaning indivisible. The word atom is derived from the Greek word atomos. Atoms are composed of three basic particles: electrons, protons, and neutrons. Electrons and protons represent the basic units of negative and positive electric charge. Atoms are electrically balanced when they have the same number of protons and electrons. The number of protons in the nucleus (core) of an atom is its atomic number. Ancient Greeks in 600 B.C. found that rubbing fur on various substances such as Amber would cause a particular attraction between the two. If they rub the Amber long enough they could even get a spark to jump. In 1600 William Gilbert an English scientist coined to the modern Latin word electricus from elektron, the Greek word for Amber.
Electrochemistry
By: Michael Barela
Electrochemistry is the study of reactions in which charged particles (ions or electrons) cross the interface between two phases of matter, typically a metallic phase (the electrode) and a conductive solution, or electrolyte. This chemical reaction generally known as an electrode process is also called electrode reactions.
In the fourth century B.C. Greek philosophers believed that matter was composed of indestructible building blocks that were so small that they could not be seen. They called these building blocks atomos, meaning indivisible. The word atom is derived from the Greek word atomos. Atoms are composed of three basic particles: electrons, protons, and neutrons. Electrons and protons represent the basic units of negative and positive electric charge. Atoms are electrically balanced when they have the same number of protons and electrons. The number of protons in the nucleus (core) of an atom is its atomic number. Ancient Greeks in 600 B.C. found that rubbing fur on various substances such as Amber would cause a particular attraction between the two. If they rub the Amber long enough they could even get a spark to jump. In 1600 William Gilbert an English scientist coined to the modern Latin word electricus from elektron, the Greek word for Amber.
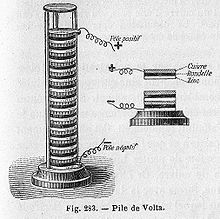
In 1793 Alessandro Volta discovered that electricity could be produced by placing too dissimilar metals on opposite sides of a moistened paper. In 1800 Nicholson and Carlisle, discovered that using Volta’s primitive battery as a source, showed that electric current could decompose water into oxygen and hydrogen. It proved to be one of the most significant experiments in the history of chemistry. This was the first proof that water is composed of hydrogen and oxygen.
This newfound discovery implied that the atoms of hydrogen and oxygen were associated with positive and negative electric charges. In 1812 the Swedish chemist Berzelius could propose that all atoms are electrified, hydrogen and the medals being positive, the non-metals negative.
This newfound discovery implied that the atoms of hydrogen and oxygen were associated with positive and negative electric charges. In 1812 the Swedish chemist Berzelius could propose that all atoms are electrified, hydrogen and the medals being positive, the non-metals negative.
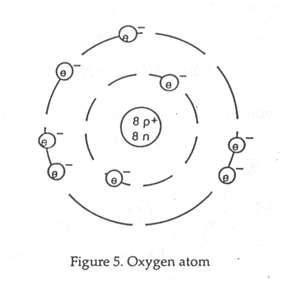
The atom contains a central core called the nucleus. The nucleus contains protons and neutrons. The number of protons in each atom equals the atomic number of that element. It also represents the number of protons in the atom. The electrons travel around the nucleus of the atom in orbital paths that are referred to as shells. The outermost shell of the atom is called the valence shell. The valence shell of a given atom cannot hold more than eight electrons. A neutral atom has an equal number of protons and electrons.
The seemingly limitless number of materials in the universe are made from different atoms called elements. Each element has its own unique atomic structure. Copper, gold, hydrogen, oxygen, and mercury are some examples of the 88 naturally occurring elements. There are additional elements that have been made artificially by bombarding atoms with high energy atomic particles. Together there are 116 elements arranged accordingly to their characteristics on a chart called a periodic table.
The smallest amount of material that can exist and still maintain the properties of that material is called a molecule. Molecules are usually made from combinations of different atoms but not always. The molecules of most gases are formed from pairs of like atoms. Materials made from combinations of different atoms are called compounds. Some common compounds are table salt, ammonia, and water.
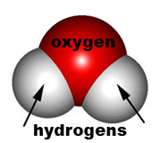
Water is unique because it is the only natural substance that is found in all three physical states on earth, solid, liquid, and gas. Pure water, number 7 on the pH scale is considered neutral. The chemical formula for water is H2O. It contains 2 hydrogen atoms for every 1 oxygen atom. Water has the ability to dissolve a wide variety of substances. A substance that dissolves when mixed with another substance is called a solute. When a solute is dissolved in a liquid, such as water, the liquid is called a solvent. Because water is polar it is a good solvent. The polarity is because oxygen is more attractive to electrons, that is it is more electronegative, than most other atoms, including hydrogen.
The smallest amount of material that can exist and still maintain the properties of that material is called a molecule. Molecules are usually made from combinations of different atoms but not always. The molecules of most gases are formed from pairs of like atoms. Materials made from combinations of different atoms are called compounds. Some common compounds are table salt, ammonia, and water.
Water is unique because it is the only natural substance that is found in all three physical states on earth, solid, liquid, and gas. Pure water, number 7 on the pH scale is considered neutral. The chemical formula for water is H2O. It contains 2 hydrogen atoms for every 1 oxygen atom. Water has the ability to dissolve a wide variety of substances. A substance that dissolves when mixed with another substance is called a solute. When a solute is dissolved in a liquid, such as water, the liquid is called a solvent. Because water is polar it is a good solvent. The polarity is because oxygen is more attractive to electrons, that is it is more electronegative, than most other atoms, including hydrogen.
Water is a powerful solvent that can facilitate chemical reactions, which are changes in the chemical composition of substances. Solutes in a mixture, called reactants, can come in contact with each other, permitting the modification of chemical bonds that occur during a reaction. The ability of elements to make chemical bonds depends on the atoms electron configuration. The electrons in the electron cloud surrounding the atoms nucleus have different energy levels based on their distance from the nucleus. The electrons located closest to the nucleus have the lowest energy, the electrons in the second shell have a little more energy, and the electrons in the third shell have even more energy. The first shell holds 2 electrons, and the second and third shells each hold a maximum of 8 electrons. When the valence shell is full, the atom will not normally form chemical bonds with other atoms. Atoms whose valence shells are not full of electrons often combine via chemical bonds. When atoms share electrons, a type of bond called a covalent bond is formed.
Atoms with 1, 2, or 3 electrons in their valence shell tend to lose electrons and therefore become positively charged ions, while atoms with 6 or 7 electrons in the valence shell tend to gain electrons and become negatively charged ions. Positively and negatively charged ions associate into a type of bond called the ionic bond. Electrically charged ions do not have an equal number of protons and electrons.
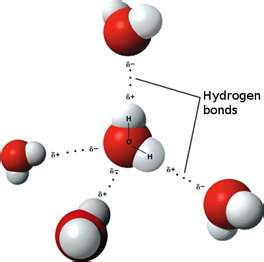
The weak attraction between hydrogen atoms and oxygen atoms in adjacent molecules forms a hydrogen bond. Hydrogen bonding is a type of week chemical bond that forms when a partially positive hydrogen atom is attracted to a partially negative atom. Water is also cohesive, it’s the tendency of like molecules to stick together and it’s called cohesion.
Like magnets, opposite electrical charges attract each other. Hydrogen atoms are partially positive, and oxygen atoms are partially negative. The hydrogen atoms attached to one side of the oxygen atom result in a water molecule where hydrogen has a positive charge and oxygen has a negative charge. Two hydrogen atoms and one oxygen atom make H2O. This is called a hydrogen bond.
Atoms with 1, 2, or 3 electrons in their valence shell tend to lose electrons and therefore become positively charged ions, while atoms with 6 or 7 electrons in the valence shell tend to gain electrons and become negatively charged ions. Positively and negatively charged ions associate into a type of bond called the ionic bond. Electrically charged ions do not have an equal number of protons and electrons.
The weak attraction between hydrogen atoms and oxygen atoms in adjacent molecules forms a hydrogen bond. Hydrogen bonding is a type of week chemical bond that forms when a partially positive hydrogen atom is attracted to a partially negative atom. Water is also cohesive, it’s the tendency of like molecules to stick together and it’s called cohesion.
Like magnets, opposite electrical charges attract each other. Hydrogen atoms are partially positive, and oxygen atoms are partially negative. The hydrogen atoms attached to one side of the oxygen atom result in a water molecule where hydrogen has a positive charge and oxygen has a negative charge. Two hydrogen atoms and one oxygen atom make H2O. This is called a hydrogen bond.
Water molecules have an overall neutral charge because they have the same number of electrons and protons. The electrons are asymmetrically distributed, which makes the molecule polar. That means it has a positive charge on one side and a negative charge on the other. The oxygen nucleus draws electrons away from the hydrogen nuclei, leaving these nuclei with a small net positive charge. The excess of electron density on the oxygen atom creates weakly negative regions at the other two corners of an imaginary tetrahedron (which means 4 sided).
Table salt is an ionic compound. It is composed of positively charged ions (base), and negatively charged ions (acid). Sodium chloride (NaCl) is one sodium atom and one chlorine atom. The sodium atom (Na) has 11 electrons. That means its valence shell has one electron. The chlorine atom (Cl) has 17 electrons. That means its valence shell has 7 electrons. The sodium atom gives up its one electron on its valence shell to the chlorine atom making the sodium positively charged in the chlorine negatively charged. The product is electrically neutral.
Naturally nature does not process matter to an excess of positive or negative charges. Matter tries to remain neutral; however we can use electrochemistry to create voltage.
If we immerse a piece of zinc metal in a solution of copper sulfate the zinc metal quickly becomes covered with a black coating of metallic copper. The reaction is an oxidation-reduction process, a transfer to electrons from the zinc to the copper. Negative electrons are removed from the zinc by copper ions that come into contact with it. The solution remains electrically neutral. In order to use the electrons we could drain them off through an external circuit that forms part of a complete electrochemical cell.
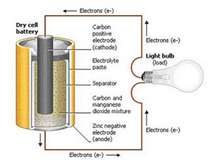
Batteries have two electrodes, an anode and a cathode. The medium that allows the electric charge to flow between electrodes is known as the electrolyte. When a load completes the circuit between the two terminals, the battery produces electricity through a series of electromagnetic reactions between the anode, cathode and electrolyte.
Table salt is an ionic compound. It is composed of positively charged ions (base), and negatively charged ions (acid). Sodium chloride (NaCl) is one sodium atom and one chlorine atom. The sodium atom (Na) has 11 electrons. That means its valence shell has one electron. The chlorine atom (Cl) has 17 electrons. That means its valence shell has 7 electrons. The sodium atom gives up its one electron on its valence shell to the chlorine atom making the sodium positively charged in the chlorine negatively charged. The product is electrically neutral.
Naturally nature does not process matter to an excess of positive or negative charges. Matter tries to remain neutral; however we can use electrochemistry to create voltage.
If we immerse a piece of zinc metal in a solution of copper sulfate the zinc metal quickly becomes covered with a black coating of metallic copper. The reaction is an oxidation-reduction process, a transfer to electrons from the zinc to the copper. Negative electrons are removed from the zinc by copper ions that come into contact with it. The solution remains electrically neutral. In order to use the electrons we could drain them off through an external circuit that forms part of a complete electrochemical cell.
Batteries have two electrodes, an anode and a cathode. The medium that allows the electric charge to flow between electrodes is known as the electrolyte. When a load completes the circuit between the two terminals, the battery produces electricity through a series of electromagnetic reactions between the anode, cathode and electrolyte.
The anode experiences an oxidation reaction in which two or more ions from the electrolyte combine with the anode, producing a compound and releasing one or more electrons. The cathode goes through a reduction reaction in which the cathode substance, ions and free electrons also combine to form compounds. The reaction in the anode creates electrons, and the reaction in the cathode absorbs them producing electricity.